Table 14.1 - Densities of Some Common Substances SolidsĪs you can see by examining Table 14. Plasma will not be discussed in depth in this chapter because plasma has very different properties from the three other common phases of matter, discussed in this chapter, due to the strong electrical forces between the charges. At high temperatures, molecules may disassociate into atoms, and atoms disassociate into electrons (with negative charges) and protons (with positive charges), forming a plasma. Definitions, online calculator and figures and tables with water properties like density, specific weight and thermal expansion coefficient of liquid water at temperatures ranging 0 to 360C (32 to 680F). There exists one other phase of matter, plasma, which exists at very high temperatures. Go watch some of Shaun Spaldings tutorials, firstly to learn the basics of GML, since you are starting out, and secondly, he has a water tutorial which may get. Water - Density, Specific Weight and Thermal Expansion Coefficients. In this chapter, we generally refer to both gases and liquids simply as fluids, making a distinction between them only when they behave differently. When placed in an open container, gases, unlike liquids, will escape. This makes gases relatively easy to compress and allows them to flow (which makes them fluids). In contrast, atoms in gases are separated by large distances, and the forces between atoms in a gas are therefore very weak, except when the atoms collide with one another. Because the atoms are closely packed, liquids, like solids, resist compression an extremely large force is necessary to change the volume of a liquid. When a liquid is placed in a container with no lid, it remains in the container. That is, liquids flow (so they are a type of fluid), with the molecules held together by mutual attraction. The mass of water is almost equal to its volume at room temperature and the water. at the temperature of 20 C to 25 C the density of the water is 0.9982 g/cc, or 998.2 kg/m 3. This occurs because the atoms or molecules in a liquid are free to slide about and change neighbors. It is concluded that the density of water is a constant value, that is, 1 g/ml or 1 g/cm 3. Liquids deform easily when stressed and do not spring back to their original shape once a force is removed. A gas must be held in a closed container to prevent it from expanding freely and escaping. (c) Atoms in a gas move about freely and are separated by large distances. Forces between the atoms strongly resist attempts to compress the atoms. (b) Atoms in a liquid are also in close contact but can slide over one another. Physical properties Water is the chemical substance with chemical formula H 2O one molecule of water has two hydrogen atoms covalently bonded to a single oxygen atom. Corn syrup, being more dense, would sink if placed in water.\): (a) Atoms in a solid are always in close contact with neighboring atoms, held in place by forces represented here by springs. 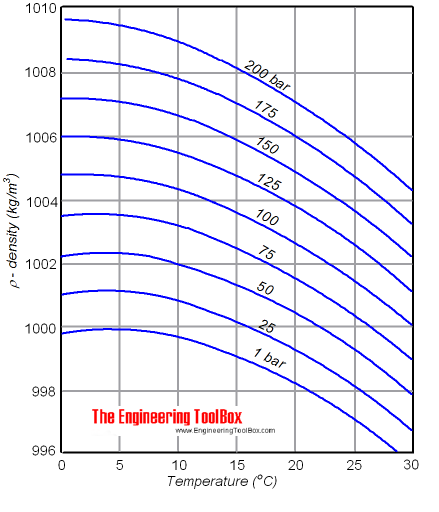
The following are the common factors that affect the density of water. This behavior of the density of water explains why ice forms at the top of a body of water. For example, water’s density under normal conditions is 0.9998395 g/ml at 4.0° Celsius (39.2° Fahrenheit), to be specific but is rounded off to 1 gram per mL or cubic centimeter (1g/mL or cc) for uniformity. 
Ice floats in water, and it can be seen from the table that ice is less dense. The density of water increases with decreasing temperature, reaching a maximum at 4.0 ☌, and then decreases as the temperature falls below 4.0 ☌. Since most materials expand as temperature increases, the density of a substance is temperature dependent, and usually decreases as temperature increases.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
